Intelect® RPW 2
Images of the RPW2 may show optional accessories not included with the 2176KIT RPW2 with standard accessories.
Please see standard accessories tab, product literature, or contact Chattanooga if you have any questions.
Click here to Download the Intelect Connect App from the Microsoft store
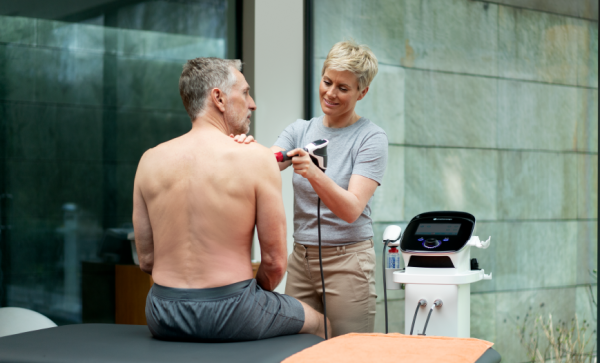
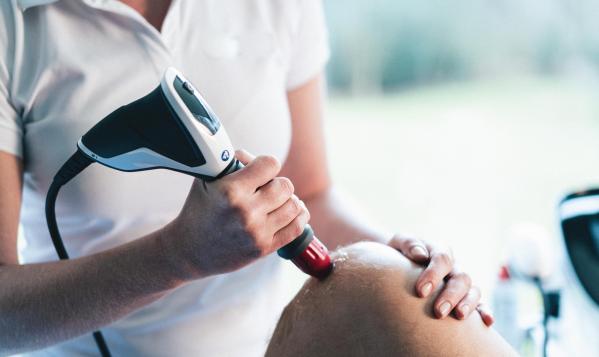
The Intelect® RPW 2 brings the next generation of shockwave therapy to the treatment room using Radial Pressure Wave technology. Designed to activate connective tissue from the surface level to difficult-to-reach pathologies, the new Intelect® RPW 2 is a modality for addressing pain and inflammation in orthopaedic conditions.
Indications:
- Reduce muscle pain and aches
- Temporarily increase blood flow
- Activate connective tissue.
Ease of Use
Incorporating high resolution touch screen technology, the Intelect® RPW 2 was created with ease of use for the clinician in mind. The clinical protocols library provides a visual anatomical and pathological library as well as treatment guidelines to aid the clinician in providing effective treatments for a large range of pathologies. The Chattanooga Intelect® Connect App provides a seamless transition of patient data from the device to the PC, allowing the clinician to produce reports and to share patient data across other Chattanooga devices. Built with ease of use in mind, the Intelect® RPW 2 includes the new ergonomic Falcon® Handpiece and convenient storage placement of device accessories.
Treat a Wide Range of Pathologies
Now available with a broader range of pressure settings and 21 unique transmitter attachments, the Intelect® RPW 2 can be customized to treat a wide range of pathologies. With guidelines on recommended transmitters for specific conditions, clinicians can address tendinopathies and myofascial trigger points as well as pain and inflammation. The versatility offered by Intelect® RPW 2 makes it the true difference maker as your next revenue generating clinical asset.
Patient Comfort
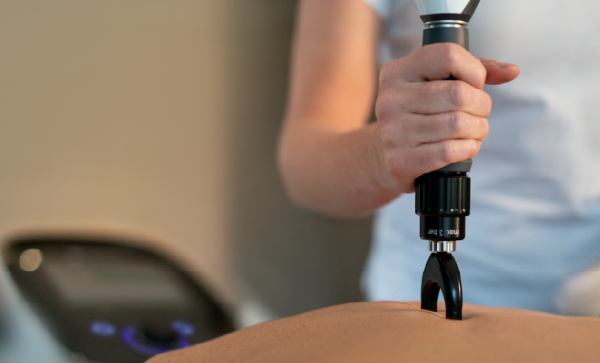
Featuring the new Falcon® Handpiece and treatment transmitters, the Intelect® RPW 2 is equipped with a broader range of bar pressures to help assist in patient comfort during treatment. Chattanooga’s proprietary ‘Comfort Mode’ provides an automatic energy ramp up, that helps create a more comfortable treatment experience for the patient.
- Conductor Transmission Gel 8.5 oz (250 ml) Bottle (24/case)
- Ro40 Beam D-Actor® Transmitter, 15mm
- D20-S Standard D-Actor® Transmitter, 20mm
- REVISION KIT FALCON® HANDPIECE SHORT SHAFT
- Power Cord
- USB Drive
| 2176KIT | 120V AC 50-60Hz 350VA |
| DIMENSIONS WXHXD | 16.15in x 16.54in x 43.31in |
| WEIGHT | 66.14 Lbs. |
| COMPRESSED AIR OUTPUT | 0.3 - 5.0 bar |
| POWER INCREMENT SETTINGS | 0.1 bar |
| PULSE AMPLITUDE | Approx 2mm at 3 bar, idling (without coupling) |
| PULSE WIDTH | Approx 6-12ms |
| PULSE FREQUENCY | FALCON® handpiece 1-21Hz for RPW, V-ACTOR® handpiece 1-50Hz for vibration |
| MODE | Single pulse or continuous |
| USER INTERFACE | 7 inch, 800x480 pixels color graphic capacitive touch screen |
| CONFORMITY | This device complies with the applicable standards EN/IEC 60601-1, CAN/CSA-C22.2 No.601.1, UL Std. No 60601-1. |
| CLASSIFICATION ACCORDING TO MDD | class IIa device 93/42 60601-1 |
LEGAL MANUFACTURER: DJO France SAS - CE 2797












![[Guide] From Research to Relief: Applying Shockwave and Laser Therapy to Common Conditions](https://www.chattanoogarehab.com/media/wordpress/705881479f255fc4ab414e9b6d05bd34.png)